Accelerate every breakthrough
Stop wrestling with trial paperwork. TrialStack creates compliant, patient‑friendly documents in minutes so your team can focus on the science.
Built on gold standards
Trusted by industry leaders for reliability and innovation
Turn scientific discoveries into treatments faster
TrialStack automates documents, ensures compliance, and keeps patients at the center so you can focus on advancing the science.
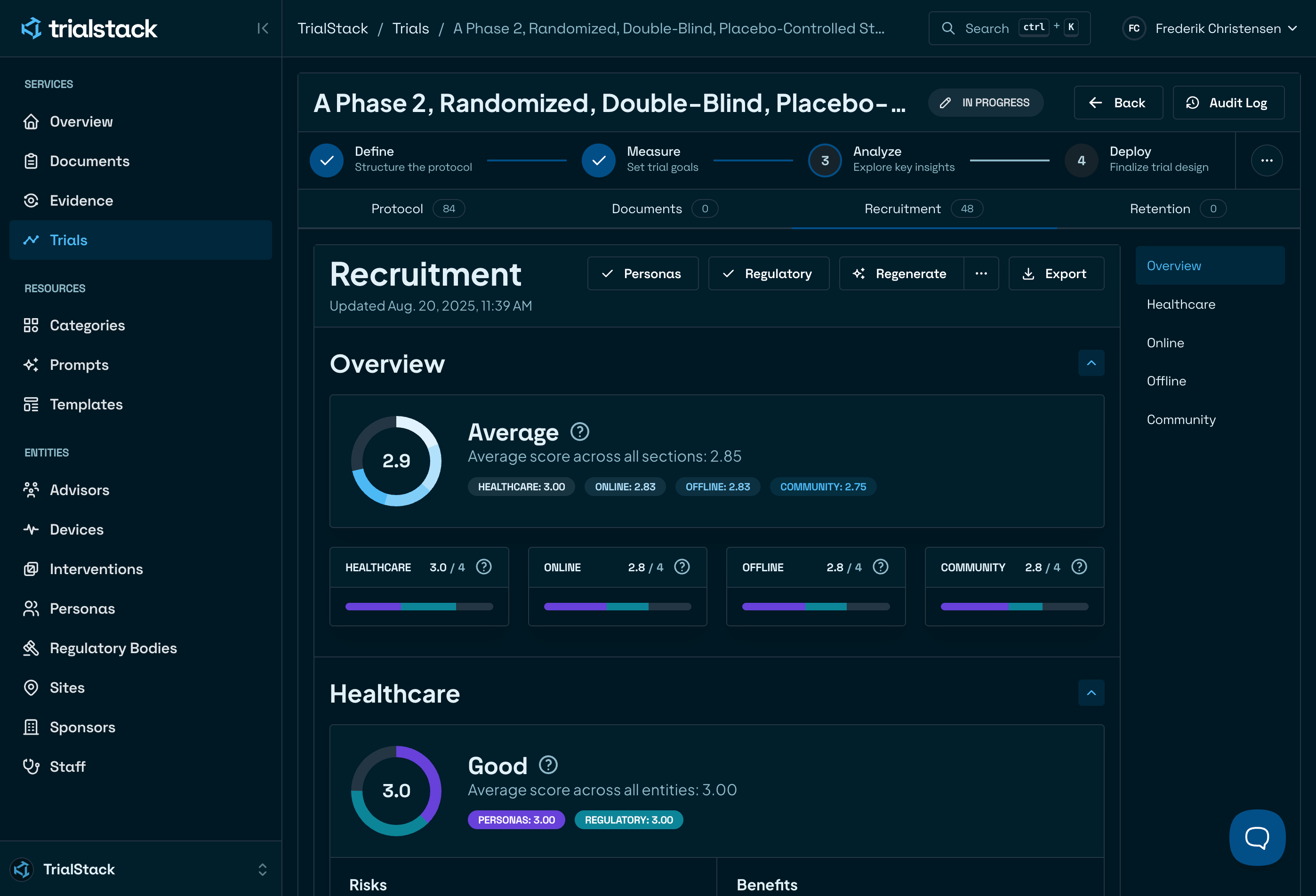
Be first to market
Automate protocols, visit schedules and patient documents to cut weeks off every study. Your team spends less time on paperwork and reaches enrollment faster.
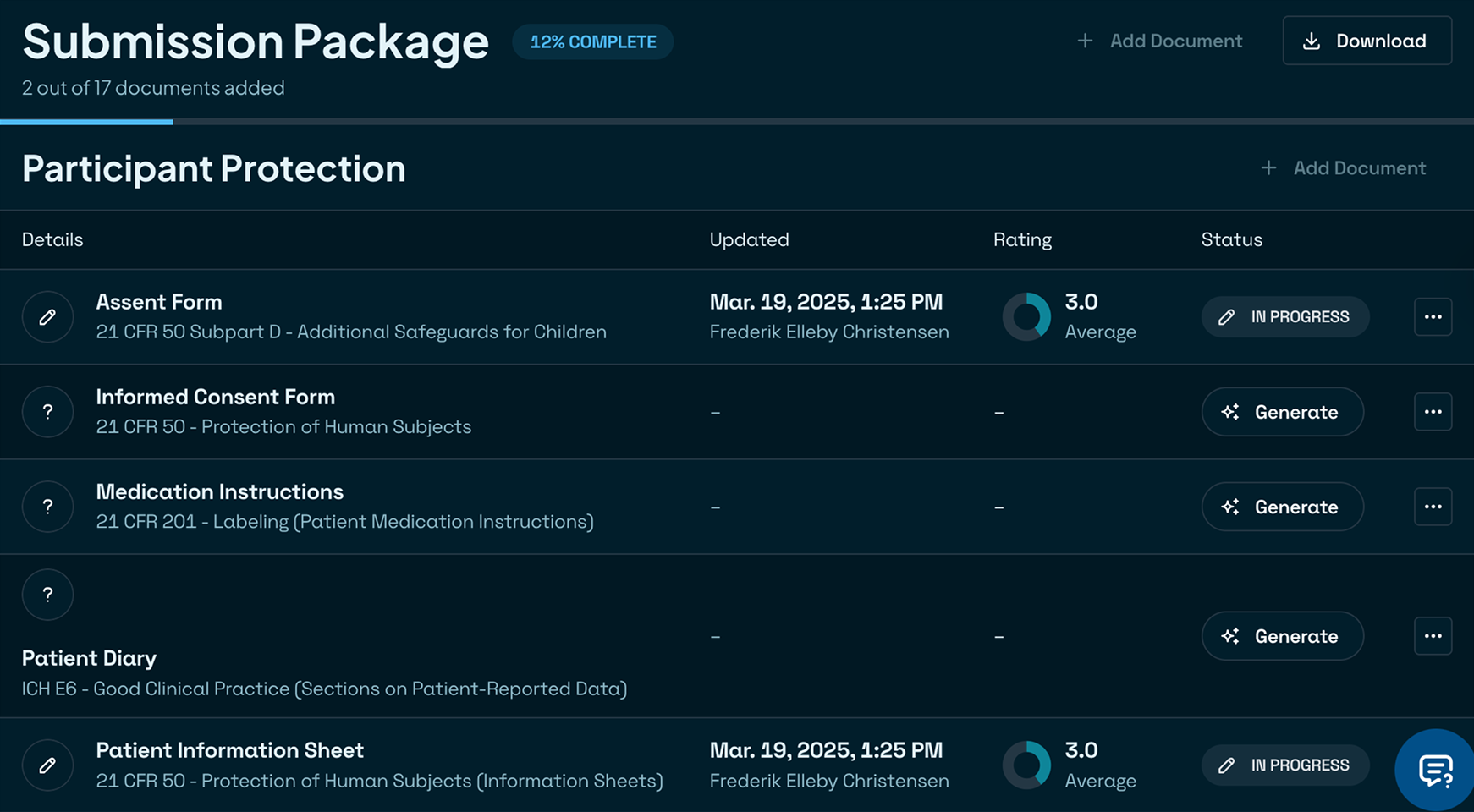
Audit-proof from day one
Built‑in regulatory rules and traceability ensure every document meets FDA/EMA and ICH standards on the first try. Rest easy knowing you’re always inspection‑ready.
Elevate your experts
Your scientists and writers focus on strategy, not formatting. TrialStack produces polished drafts so your team can spend time on high‑value decisions.
Eliminate bottlenecks
Real‑time editing and built‑in feedback loops keep every stakeholder aligned. Decisions move forward quickly instead of sitting in someone’s inbox.
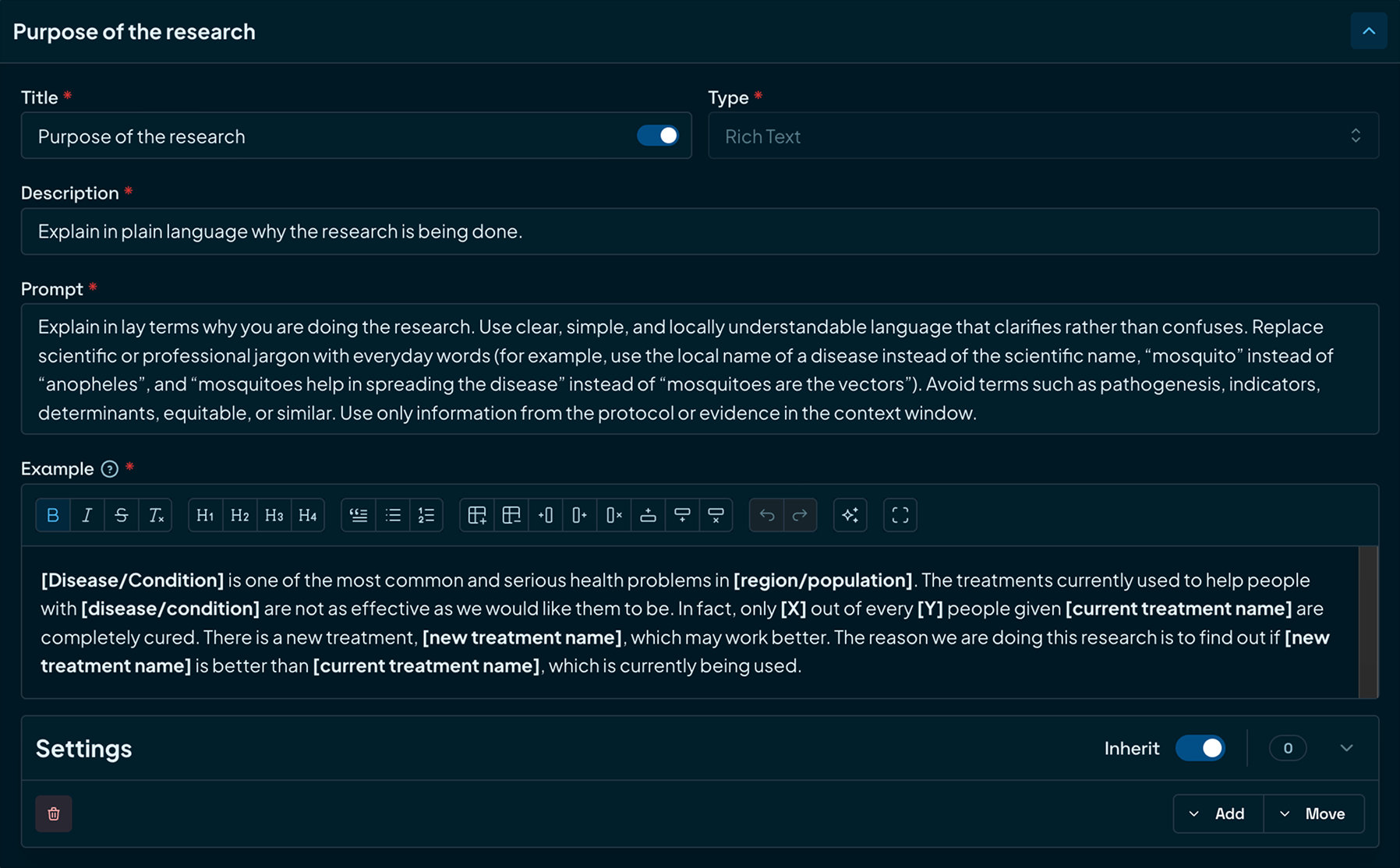
Design with patients in mind
Infuse real‑world patient insights into every study design. Create clear, engaging patient documents that improve participation and retention.
Proof Builds Trust
TrialStack is measured against the toughest standards in science and regulation
First-pass submission acceptance
Less time spent drafting documents
Regulatory accuracy you can trust
Launch compliant, patient‑centric trials faster
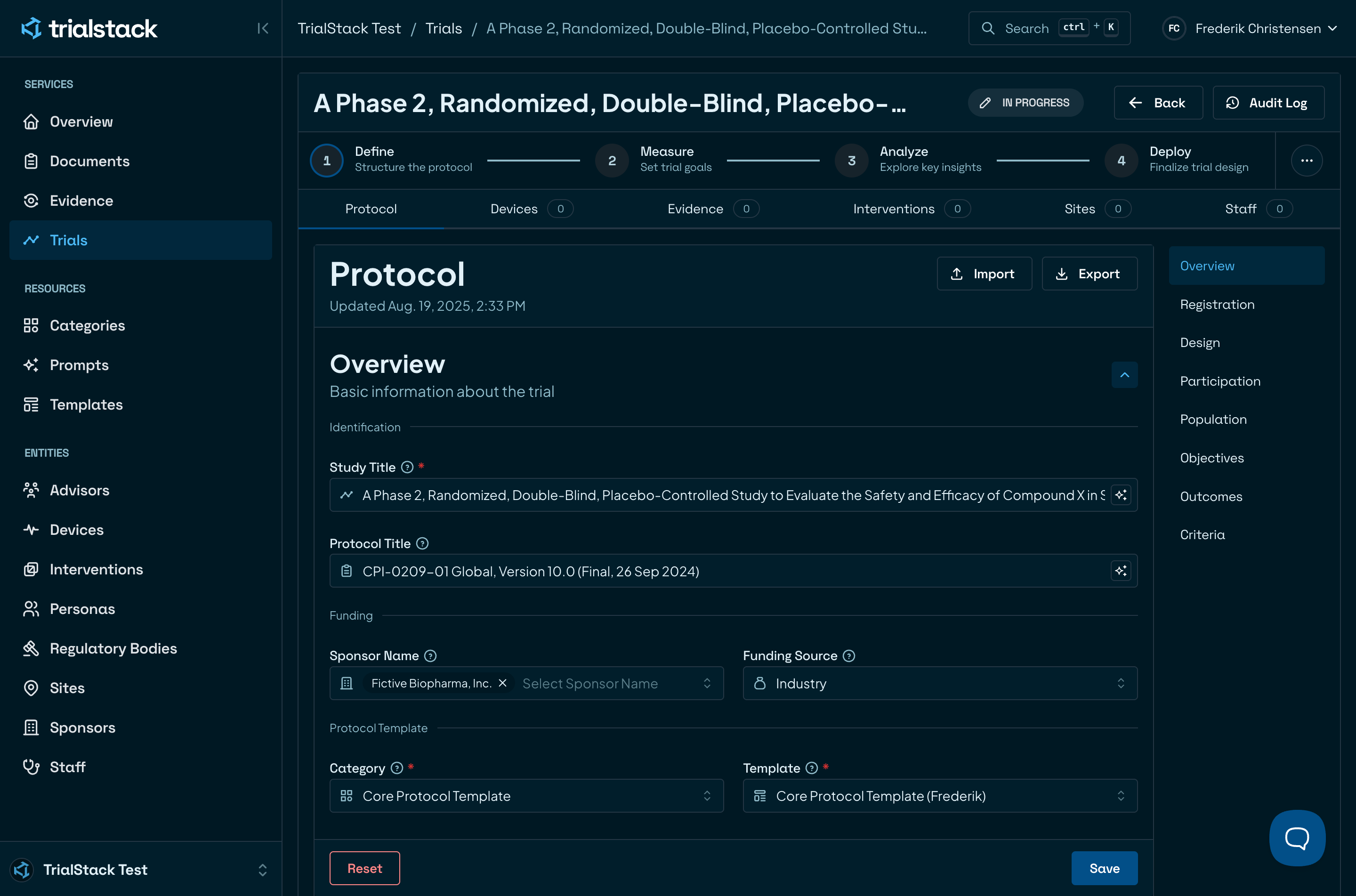
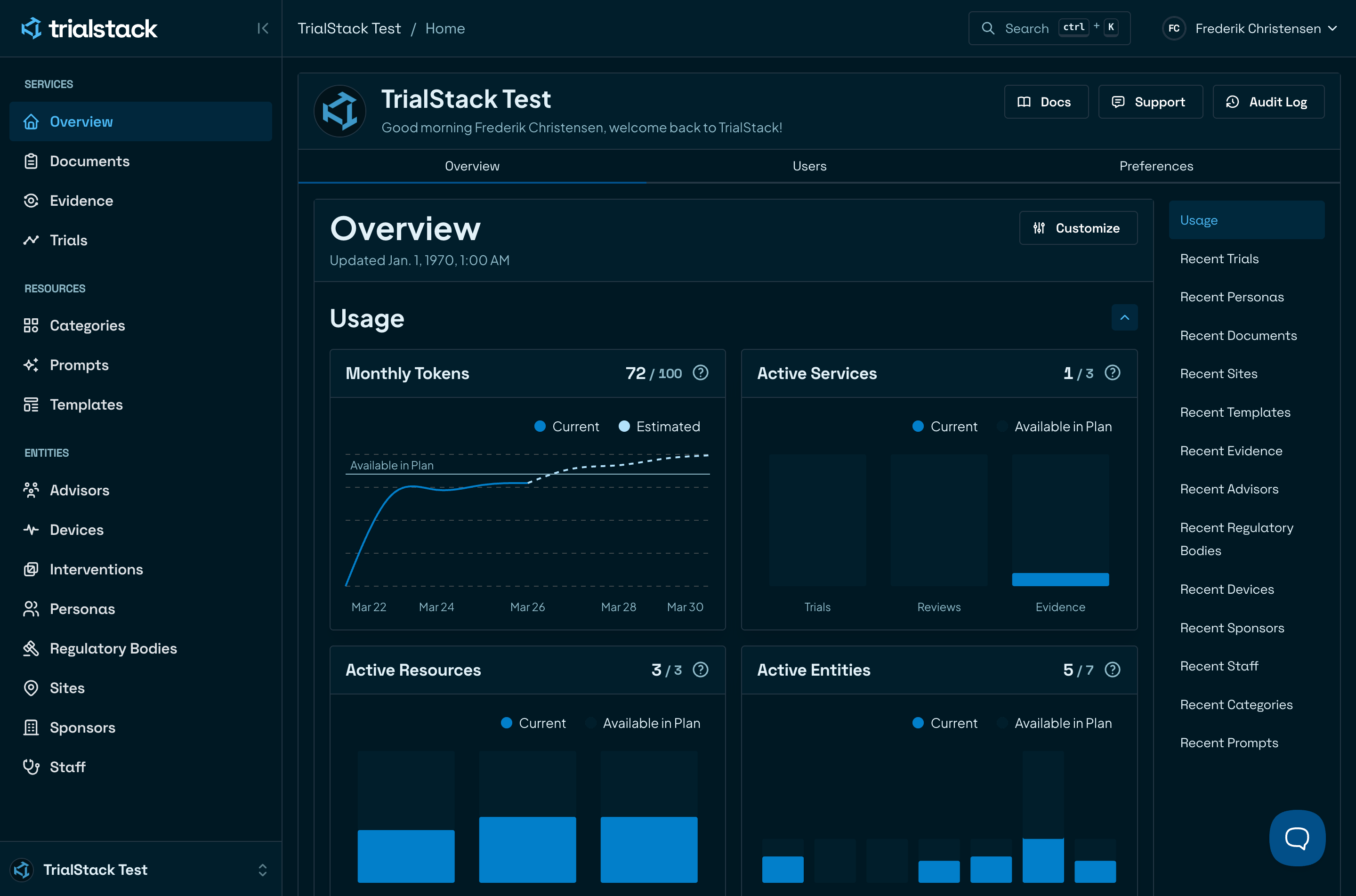
Compose every trial from one platform
Eliminate chaos with automation that organises and verifies every element for speed and compliance.
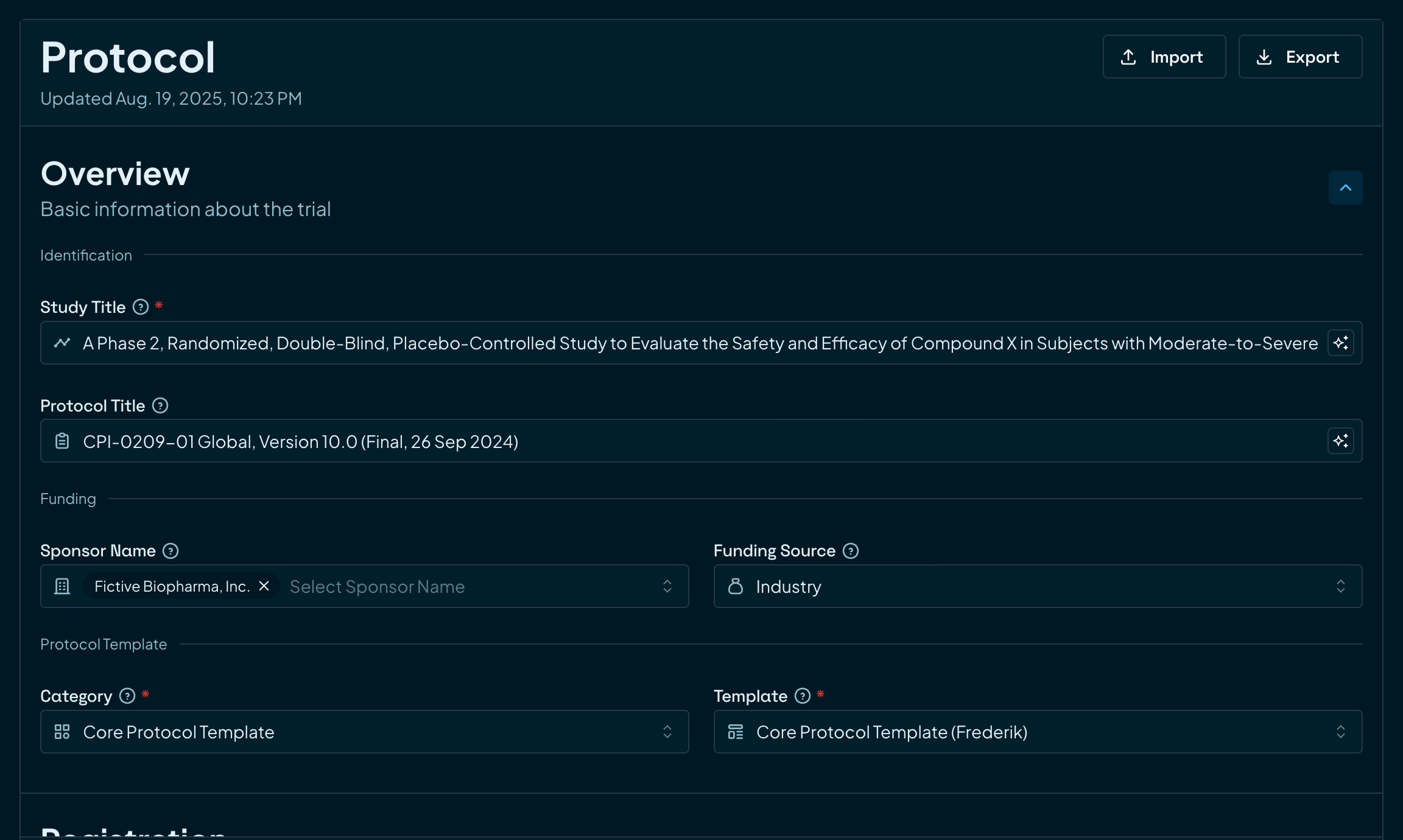
Lay a solid foundation
Start every study with structured, reliable data and clearly defined trial components. No guesswork, no missing pieces.
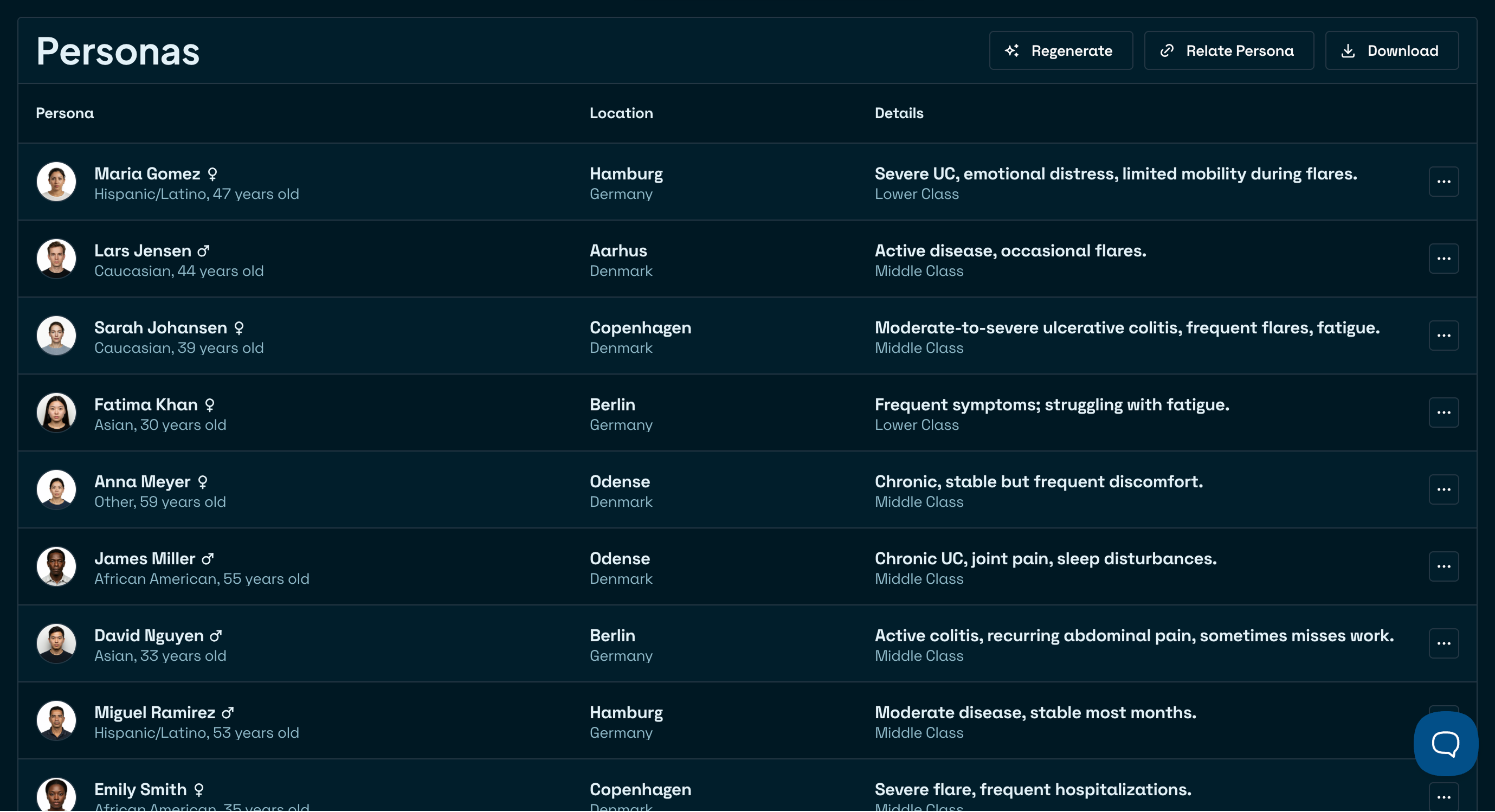
See the full picture
Understand how every element of your study fits together. Easily adapt as your trial evolves.
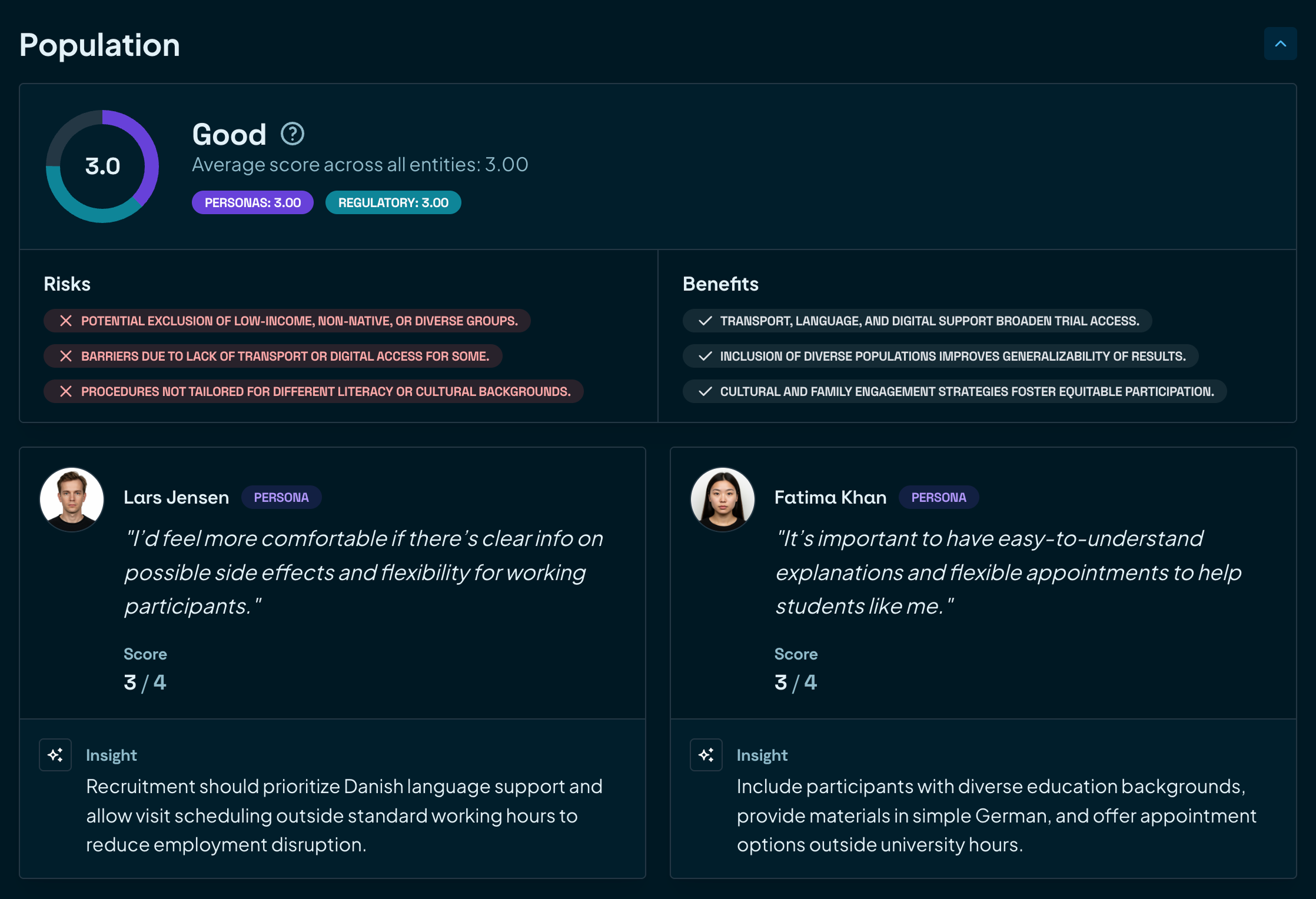
Turn feedback into action
Aggregate feedback and data to drive improvements. Remove friction and continuously enhance your trial design.
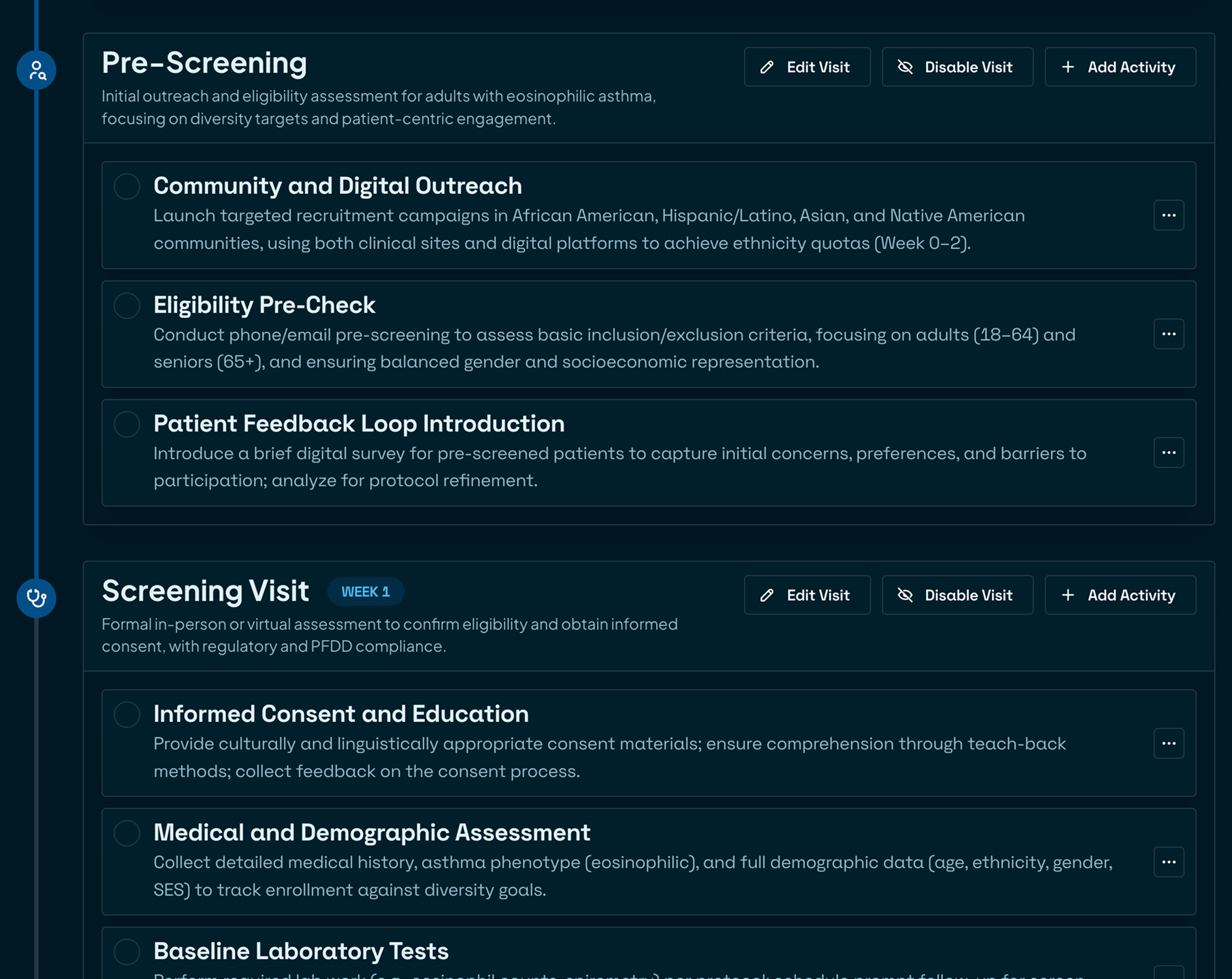
Finish with confidence
Export submission‑ready documents quickly. Comprehensive checks ensure no detail slips through the cracks.
End‑to‑end trial orchestration
Bring every aspect of your clinical trial onto one unified platform built for regulatory excellence.
Advisors
Use proven templates and patient‑centric design principles to build comprehensive protocols quickly.
Categories
Easily manage sites, investigators, and staff with intuitive tools and built‑in communication.
Devices
Coordinate devices, data sources, and site equipment seamlessly.
Files
Monitor patient enrollment, visits, and events in real time—never miss a deadline.
Interventions
Track and adjust interventions with ease, ensuring protocol compliance across all sites.
Personas
Build trials around participant personas and design with patients in mind.
Prompts
Leverage AI‑driven suggestions to improve protocol design and reduce risk.
Regulatory Bodies
Meet the requirements of every authority without rework. Our system aligns to global standards.
Sites
Simplify site selection and management with full visibility into performance.
Templates
Use customizable, best‑practice templates to jump‑start every document.
Sponsors
Give sponsors complete oversight and confidence in your documentation process.
Staff
Empower staff to collaborate, document, and report with speed and accuracy.
A GxP‑validated platform built for clinical research
Every AI feature is validated, audited, and monitored to meet ICH E6(R3), EMA reflections on AI, FDA guidance and the EU AI Act.
Risk‑based validation
Our development follows risk‑based quality management. Each AI feature has a defined context of use and validation.
Continuous testing
Software is tested end‑to‑end throughout development, with documented evidence for inspection.
Responsible AI
Built to comply with FUTURE‑AI principles for fairness, traceability, robustness and explainability.
Enterprise security
Hosted on Microsoft Azure with SOC 2, ISO 27001, HIPAA and GDPR certifications inherited.
Human oversight
AI assists but never replaces expert judgment. Performance is monitored, and users remain in control.
Global compliance
Designed to meet high‑risk AI requirements under the EU AI Act and FDA AI credibility framework.
Frequently Asked Questions
TrialStack is an AI‑driven platform that automates clinical trial documentation and workflow. It helps reduce timelines, improve quality, and ensure compliance with global standards.
TrialStack incorporates regulatory rules and templates out of the box. It supports CDISC standards, FDA’s 21 CFR Part 11 and ICH guidelines, with built‑in audit trails and validation.
TrialStack supports studies from Phase 0 through Phase 4, across therapeutic areas. It handles single‑site pilot studies and multi‑site, multinational trials alike.
Implementation depends on complexity, but simple setups can go live in 2–4 weeks. Larger deployments may take 6–8 weeks. We manage onboarding so you can start seeing benefits quickly.
We offer comprehensive onboarding, live training, documentation, and 24/7 technical support. Our customer success team ensures you’re getting full value from TrialStack.
Pricing depends on your usage—number of studies, sites and users. We offer both subscription plans and per‑study pricing. Contact sales for a custom quote.

